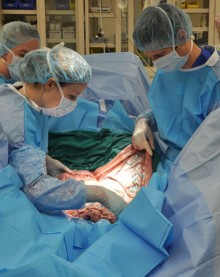
CALEC surgery, or cultivated autologous limbal epithelial cell surgery, is a groundbreaking procedure making waves in ocular regenerative medicine. This innovative treatment, pioneered at Mass Eye and Ear, leverages the body’s own limbal epithelial cells to repair the cornea and restore vision that was once deemed irreparable. With the ability to effectively treat severe eye injuries and deficiencies through stem cell therapy, current clinical trials have demonstrated impressive success rates. Patients suffering from conditions like limbal stem cell deficiency can benefit from this advanced therapy, bringing new hope for pain relief and improved visual acuity. As the field of eye injury treatment evolves, CALEC surgery stands as a testament to the potential of modern science in restoring sight and enhancing quality of life for those affected.
Cultivated autologous limbal epithelial cell surgery, often referred to as CALEC surgery, heralds a new era in treatment options for individuals with corneal damage. By utilizing a patient’s own healthy eye tissue to generate a graft, this innovative surgical technique provides a personalized approach to healing, particularly in the realm of ocular regenerative therapies. The focus on limbal epithelial cells highlights the critical role stem cells play in maintaining ocular health, especially when faced with severe injuries or debilitating conditions. This advanced procedure signifies an important step forward in eye care, aiming not only to restore vision but also to improve the overall quality of life for patients suffering from eye-related ailments. As research and technology continue to drive breakthroughs in this field, the hope for effective treatments grows ever stronger.
Introduction to CALEC Surgery
The introduction of CALEC surgery marks a significant milestone in the field of ocular regenerative medicine. Developed at Mass Eye and Ear, this innovative procedure utilizes cultivated autologous limbal epithelial cells to rejuvenate the cornea’s surface in patients suffering from severe corneal damage. As stem cell therapy continues to evolve, CALEC surgery represents a beacon of hope for those faced with debilitating eye injuries that were previously deemed irreparable. According to principal investigator Ula Jurkunas, the success of CALEC surgery could redefine treatment options for acute corneal injuries.
This pioneering clinical trial involved 14 participants and showcased results that surpassed expectations, with over 90% efficacy in restoring corneal integrity over an 18-month period. The surgery involves extracting healthy limbal epithelial cells from an unaffected eye, expanding them into a graft through a meticulous manufacturing process, and transplanted into the damaged eye. The results offer new insights into eye injury treatment methodologies, advancing the landscape of regenerative therapies aimed at restoring vision.
The Science Behind Limbal Epithelial Cells
Limbal epithelial cells are critical for maintaining the smooth surface of the cornea, ensuring optimal vision and eye health. These cells act as a protective barrier and play a crucial role in healing the eye after injury. In cases of corneal damage—like chemical burns or infections—there can be a depletion of these essential stem cells, leading to limbal stem cell deficiency. This condition not only impairs vision but also causes persistent discomfort and can severely impact patients’ quality of life.
Understanding the biology of limbal epithelial cells is pivotal for advancements in ocular regenerative medicine. Efforts at Mass Eye and Ear, led by experts like Reza Dana, have explored new avenues to regenerate these cells successfully. The approach not only emphasizes the essential role of these stem cells in corneal health but also creates a pathway for patients with bilateral injuries to potentially receive treatment in the future. Through ongoing research, the hope is to refine methods for cellular restoration and enhance patient outcomes significantly.
Innovative Manufacturing Process of CALEC Grafts
At the heart of CALEC surgery lies a groundbreaking manufacturing process for creating limbal epithelial cell grafts. This process initiates with a biopsy from a healthy eye, followed by the meticulous culturing of these cells into a viable graft. The unique procedure developed at Mass Eye and Ear has garnered attention not only for its safety but also for its effectiveness in restoring vision. The cultivation takes approximately two to three weeks, during which the cells are expanded to form a graft suitable for transplantation into the damaged eye.
This innovation represents a significant leap forward in eye injury treatment. The careful coordination between Ula Jurkunas’ surgical team and the cell manufacturing facility at Dana-Farber has demonstrated that CALEC grafts can meet stringent quality standards necessary for human transplantation. Continual enhancements in this manufacturing process are essential to address the needs of patients, particularly in scaling the approach for broader applications in ocular medicine.
Clinical Trial Results and Implications
The results of the CALEC clinical trial have offered promising insights into the future of ocular therapies. With a remarkable success rate, approximately 93% of patients experienced significant improvements in corneal restoration within 12 months. The implications of these findings are far-reaching, signaling a transformative shift in treatment paradigms for patients previously considered beyond help. As the research progresses, it paves the way for developing standardized treatments that leverage stem cell therapy for broader applications.
Moreover, the trial’s outcomes underscore the importance of rigorous research and collaboration among top institutions, like Mass Eye and Ear and Dana-Farber, which enrich the field of ocular regenerative medicine. As researchers aim for federal approval, the hope is to offer CALEC surgery to a wider population, extending this potentially life-altering treatment to individuals suffering from corneal injuries.
Safety Profile of CALEC Surgery
Safety is paramount in any surgical procedure, and CALEC surgery has shown an excellent safety profile during the clinical trial. Participants experienced no severe adverse events related to the procedure, other than mild and manageable issues. A notable case involved a bacterial infection that occurred in one participant due to pre-existing chronic contact lens use, illustrating the need for careful patient selection and postoperative care.
This favorable safety profile bolsters the credibility of CALEC surgery as a viable option for eye injury treatment. The absence of serious complications reinforces the therapy’s potential as a groundbreaking approach within ocular regenerative medicine. Such results not only instill confidence in the treatment but also encourage future researchers to explore this innovative pathway for restoring vision.
Barriers to Accessibility and Future Innovations
Despite the promising outcomes from the CALEC trial, accessibility remains a barrier to patient treatment. Currently, the procedure is still experimental and not widely available in hospitals across the U.S. The requirement for having only one healthy eye from which to harvest limbal stem cells limits the number of candidates for this potentially life-changing treatment.
Looking ahead, the goal is to develop an allogeneic manufacturing process that would utilize stem cells from cadaveric donor eyes, significantly increasing accessibility for patients with bilateral corneal damage. Researchers are optimistic that such innovations will not only improve the availability of CALEC surgery but also lead to its adoption as a standard treatment for serious ocular injuries, thus revolutionizing care within the field of ocular regenerative medicine.
The Role of FDA and Regulatory Approvals
The collaboration with the U.S. Food and Drug Administration is crucial in turning the successful results of the CALEC trial into a widely accepted treatment. Currently, the procedure is classified as experimental. The journey towards FDA approval is a detailed process involving extensive data collection, safety evaluations, and efficacy studies.
Securing regulatory approval is essential not only for this particular treatment but also for establishing a framework for future ocular regenerative medicine therapies. The CALEC study stands out as the first human trial of a stem cell therapy funded by the National Eye Institute, setting a precedent for subsequent research initiatives. Achieving FDA approval would mark a monumental step forward for patients with cornea injuries, granting them access to state-of-the-art treatments.
Collaboration and Research Partnerships
The success of the CALEC trial depended significantly on collaborative efforts among research institutions, underscoring the importance of teamwork in the medical field. Partnerships between Mass Eye and Ear, Dana-Farber, and various other contributors highlight the complex nature of developing advanced therapies. This cooperation not only streamlines the research process but also combines expertise across various disciplines, fostering innovation.
As the field of ocular regenerative medicine continues to evolve, ongoing collaborations will play a vital role in enhancing treatment protocols and expanding the therapeutic toolkit available to clinicians. Future studies involving larger cohorts and multi-center trials will further establish the safety and efficacy of CALEC surgery and may lead to breakthroughs in ocular health.
Patient Advocacy and Education Initiatives
With advancements like CALEC surgery, patient advocacy and education become increasingly essential. A significant part of bringing innovative treatments to the forefront involves ensuring that patients are well-informed about their options, treatment processes, and outcomes. Educational initiatives can empower patients with knowledge, helping them engage in conversations with healthcare providers about new therapies and clinical trials available.
Raising public awareness about the possibilities offered by stem cell therapies in the treatment of eye injuries can also cultivate a supportive environment for further research. Advocacy groups can play a pivotal role in lobbying for greater funding, creating awareness around clinical trials, and promoting access to cutting-edge treatments like CALEC surgery for individuals in need.
Looking Ahead: The Future of Ocular Regenerative Medicine
The journey of CALEC surgery is just the beginning of a broader revolution in ocular regenerative medicine. As research continues, the potential for stem cell therapies in treating various eye conditions expands significantly. Researchers are optimistic about developing novel approaches and refining existing techniques, hoping to treat a wider array of corneal injuries effectively.
Future investigations will not only aim for FDA approvals but will also seek to understand the long-term impacts of CALEC surgeries and similar therapies on visual acuity and quality of life for patients. By nurturing innovative research pathways and promoting collaboration among institutions, there is hope for a future where vision restoration is a standard achievable outcome for patients with severe ocular injuries.
Frequently Asked Questions
What is CALEC surgery and how does it help with eye injury treatment?
CALEC surgery, or cultivated autologous limbal epithelial cells surgery, involves extracting stem cells from a healthy eye to create a graft that can restore the damaged cornea. This innovative eye injury treatment has shown over 90% effectiveness in restoring corneal surfaces, providing new hope for patients who suffered from blinding corneal injuries.
How does corneal stem cell therapy work in CALEC surgery?
Corneal stem cell therapy is the cornerstone of CALEC surgery. The process begins with a biopsy of healthy limbal epithelial cells from one eye, which are then cultivated into a graft. This graft is transplanted into the damaged eye, leading to regeneration of the corneal surface and alleviating visual difficulties and pain caused by limbal stem cell deficiency.
What role do limbal epithelial cells play in ocular regenerative medicine?
Limbal epithelial cells are essential for maintaining the cornea’s surface; they help protect and regenerate the cornea. In ocular regenerative medicine, these cells are utilized in CALEC surgery to treat severe eye injuries by restoring the cornea’s integrity and function, making it a vital element of modern ophthalmic treatments.
Is CALEC surgery currently available at Mass Eye and Ear?
Currently, CALEC surgery is still considered experimental and not yet offered at Mass Eye and Ear or any other U.S. hospitals. However, ongoing clinical trials aim to gather more data to potentially secure FDA approval for this innovative procedure in ocular regenerative medicine.
What are the outcomes of the clinical trials for CALEC surgery?
In the clinical trials for CALEC surgery, about 93% of participants showed complete or partial restoration of the cornea after 18 months. Results indicated that the procedure significantly improved patients’ vision and demonstrated a high safety profile, with no serious complications reported.
What future advancements are expected in CALEC surgery?
Future advancements in CALEC surgery may include establishing an allogeneic manufacturing process for limbal stem cells from cadaveric donor eyes. This development could expand treatment options to patients with damage to both eyes, thereby broadening the impact of this revolutionary eye injury treatment.
Are there any risks associated with CALEC surgery?
While CALEC surgery has displayed a high safety profile, the main risks include minor adverse events; one participant experienced a bacterial infection related to chronic contact lens use. As the procedure is still in the experimental phase, ongoing monitoring is essential to ensure patient safety during clinical trials.
| Key Points |
|---|
| Ula Jurkunas performs the first CALEC surgery at Mass Eye and Ear. |
| CALEC stands for Cultivated Autologous Limbal Epithelial Cells, a stem cell therapy for corneal injuries. |
| The clinical trial involved 14 patients over 18 months showing over 90% effectiveness. |
| The method involves harvesting stem cells from a healthy eye, expanding them, and transplanting to a damaged eye. |
| Previous treatments for severe corneal injuries were limited; CALEC offers new hope for those previously deemed untreatable. |
| The FDA approved the clinical trial, marking it as a significant breakthrough in eye care. |
| The results showed 93% overall success at the 12-month mark with varying improvement in vision among participants. |
| Future improvements may include using donor eyes for patients with bilateral damage, expanding treatment options. |
| Research is ongoing, and more trials are needed for potential FDA approval, showing promise in ophthalmology. |
Summary
CALEC surgery represents a groundbreaking advancement in the treatment of corneal injuries, significantly improving the prospects for patients who suffer from injuries previously considered untreatable. This innovative stem cell therapy has shown remarkable success rates in clinical trials, paving the way for future developments in eye care. By harnessing stem cells from healthy eyes, CALEC surgery helps restore corneal surfaces effectively, offering hope to those affected by blinding conditions. As we look forward to further research and potential FDA approval, CALEC surgery stands out as a beacon of progress in regenerative medicine.